Alopecia areata, an autoimmune disease where the body attacks its own hair, is a common condition that affects approximately 2.5 million people in the United States, with men and women suffering equally. The condition has made its way into pop culture recently, with stars like Jada Pinkett Smith and Ricki Lake bringing attention to the topic. While there are currently no treatments approved for the condition, some companies are getting close to a solution.
On May 23, Concert Pharmaceuticals announced that its experimental treatment for alopecia areata showed promising results in a clinical trial. The twice-daily pill, CTP-543, was put to the test in a study of about 700 people with moderate to severe alopecia areata who had lost at least half of their hair, with the final group, on average, having less than 16 percent of their hair left, reported The Boston Globe. The recent trial found that the pill could restore almost a full head of hair (80 percent of their scalp covered) in 40.5 percent of patients in the 12 mg twice-daily group and 30 percent of patients in the 8 mg twice-daily group in 24 weeks.
Dr. Lynne J. Goldberg, director of the hair clinic at Boston Medical Center and professor of dermatology at Boston University School of Medicine, told The Boston Globe that she’s optimistic that when using the drug for more than 24 weeks, even more patients will see impressive regrowth. “I don’t think 40 percent will be the full number. Some people will take longer to respond,” she said.
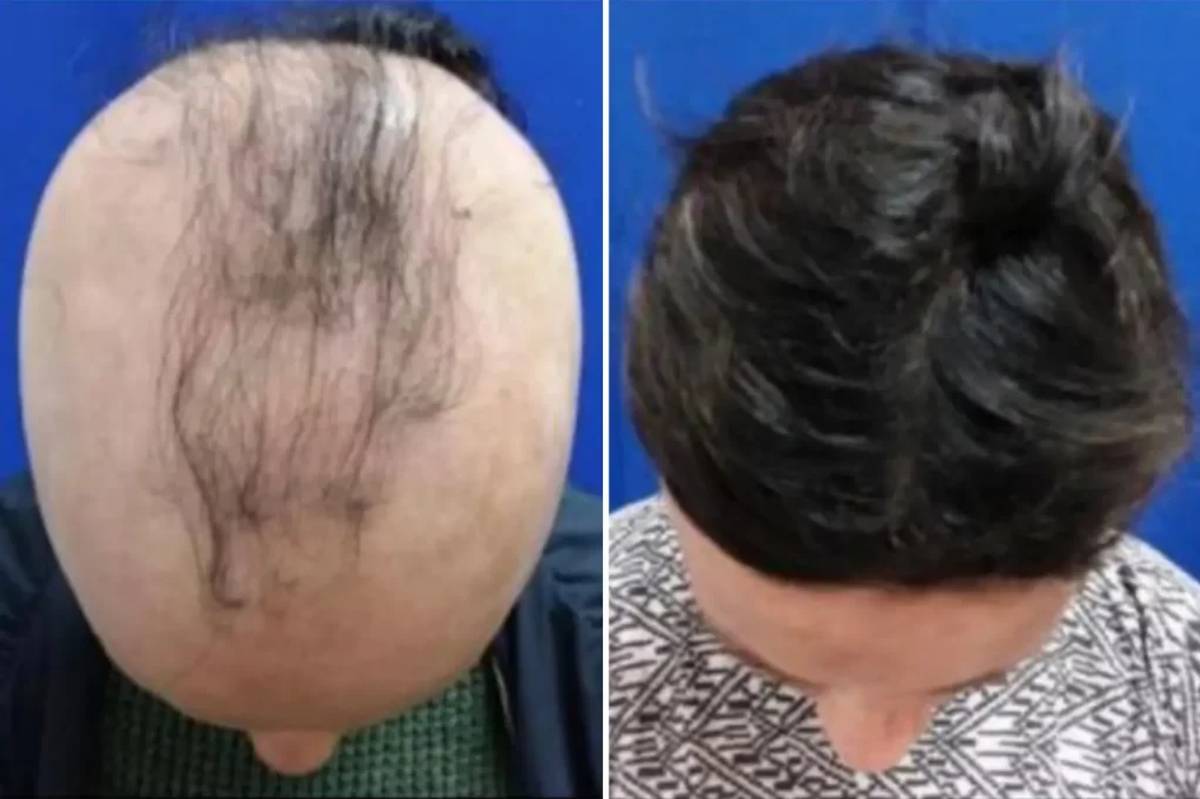
Concert Pharmaceuticals
“Today marks an important milestone in advancing new treatments for alopecia areata, and I’m so happy to see such positive results from the first Phase 3 trial with CTP-543,” Brett King, MD, Department of Dermatology, Yale University School of Medicine and clinical investigator in the trial said in the statement. “There is a great need for treatments for this challenging disease, and the results from the [the] trial suggest that CTP-543 may potentially provide an important therapy for treating alopecia areata.” According to the statement, they plan to submit a New Drug Application to the U.S. Food and Drug Administration in the first half of 2023.